Synergy Diagnostic Laboratory, Inc.
The Foundation — Davie, Florida — Est. 2012
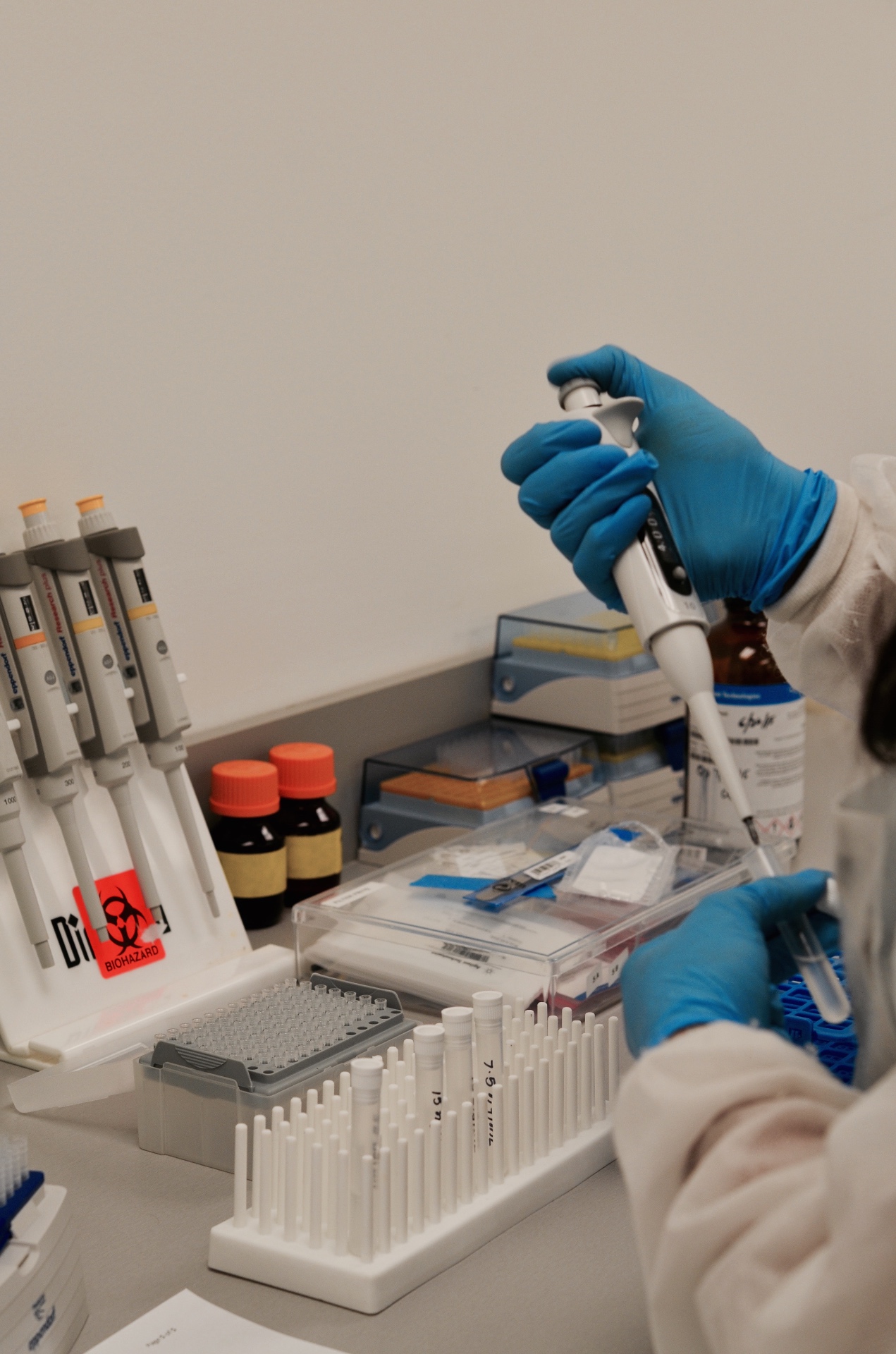
The flagship operation and heart of the Synergy ecosystem. A CLIA Certified, COLA Accredited High Complexity Laboratory credentialed with Medicare nationally, Medicaid across 30 states, Humana, Tricare West, and Humana Military / Tricare East. Licensed to operate in all 50 states and Puerto Rico.
Built around a fleet of 10+ Agilent Technologies LC-MS/MS systems — one of the largest independent clinical toxicology platforms in the Southeast — Synergy served pain management physicians, addiction treatment centers, and clinical practices across the country with fast, accurate, and defensible results at scale.
When COVID-19 emerged, Synergy rapidly deployed a PerkinElmer automated molecular platform capable of processing 3,100 specimens per day — earning two FDA Emergency Use Authorizations under its own name, a distinction achieved by very few independent laboratories in the United States.
"One Drop Technology"